Product Description
Rapid Single/Multi drug Test is a rapid, screening test for the qualitative detection of single/multiple drugs and drug metabolites in human urine at specified cut off levels.For professional use. For in vitro diagnostic use only.
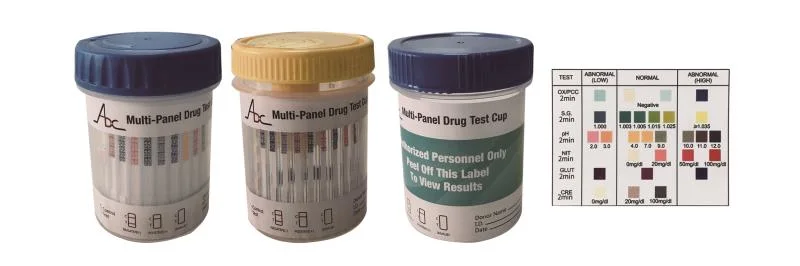
Product Characteristic
Specimen: Urine
Time to result: 1-5 minutes
Shelf Time: 24 Months ( 2 years )
Accuracy:> 99%
Storage: 4°C - 30°C
Certificates:CE, ISO 13485
Product Features
Faster result
Less sample volume
Results stable up to 30 minutes
Company Profile
Established in Feb.2019, Anhui Formaster Biosci Co., Ltd. is located in Anhui Wuwei Economic Development Zone. The site area is 1,500 sq. meters with a clean room of 300 sq. meters. The company is specialized in researching, developing and manufacturing in vitro diagnostic (IVD) devices with advanced technology, high R&D input and innovation. Granted CE certification, the leading products are rapid diagnostic devices, focusing on the following 8 categories, covIng Antigen, coving antibody, women's health, infectious diseases, drugs, cancer, cardiovascular and respiratory. Our professional team is the national leader in colloidal gold, latex and other labelling technologies, biological raw materials technologies, including monoclonal antibodies, polyclonal antibodies, genetic recombinant antigens, synthetic antigen, etc., as well as other cutting-edge biological fields, such as immunochromatography.Our latest product, New C0R0NAVIRUS C0VID IgG / IgM Rapid Test Device, is ISO 13485:2016 certified, registered in MHRA, UK and listed in Medical Devices and Supplies Companies with Certification/Authorization from other Countries (CCCMHPIE, China) for exporting and selling worldwide (not including China mainland). Our star product, New C0R0NAVIRUS C0VID Antigen Rapid Test Device (swab) is CE marked with a capacity of detecting coving antigens at the concentration of 100 pg/mL.



Certificates





